Ampoule ISO Standard YBB Standard High Quality
- Supplier:
 Triumph Junheng Co.,Ltd
Triumph Junheng Co.,Ltd
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- 100000 pc
- Supply Capability:
- 1000000000 pc/month
OKorder Service Pledge
OKorder Financial Service
You Might Also Like
Ampoule ISO Standard YBB Standard High Quality

Neutral borosilicate glass is far superior to low borosilicate glass in terms of hydrolitic resistance, acid and alkali resistance, freezing resistance, filling speed,etc. and it is not easy to cause delamination"due to chemical soaking and corrosion. It is an internationally recognized safe pharmaceutical packaging materials.
Ampoule YBB Standard
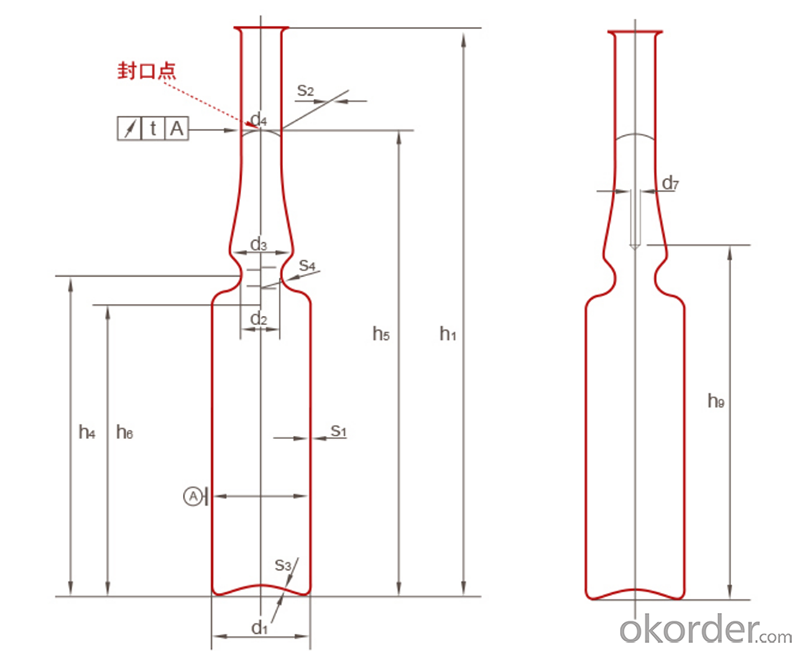
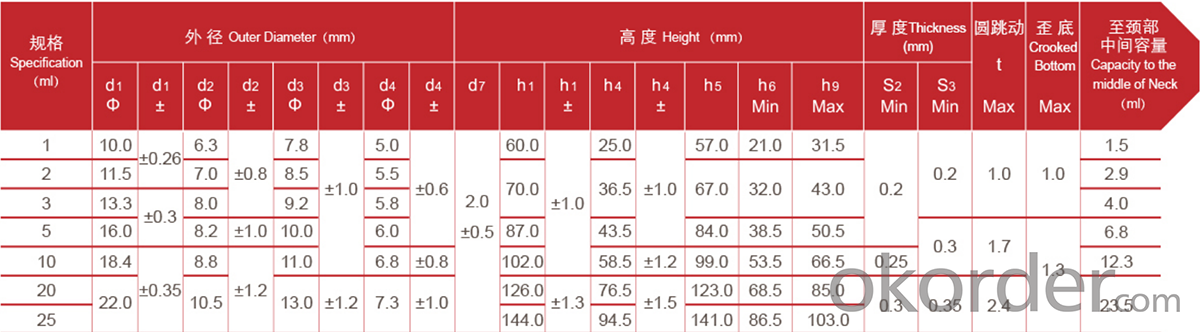
Ampoule ISO Standard
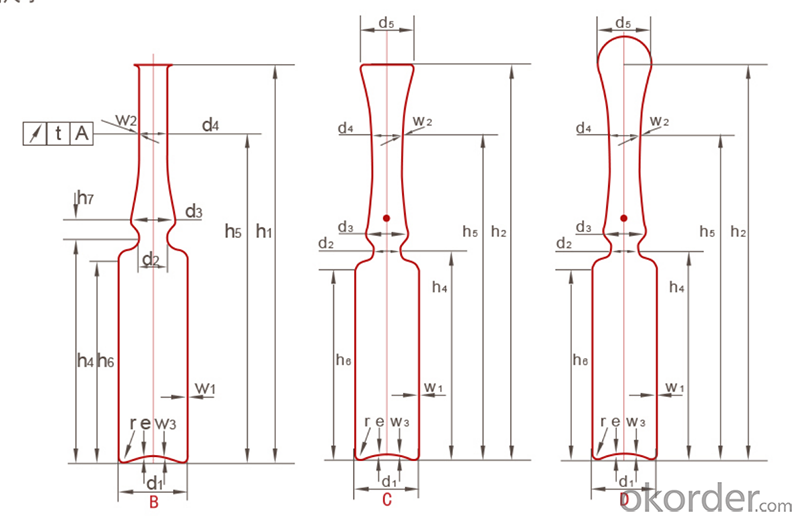
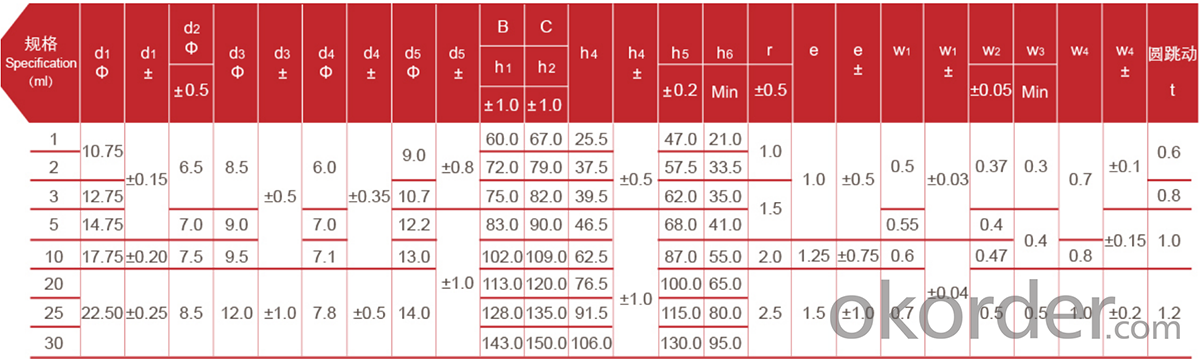

The converting products workshop of the plant has a 100000-level clean room that meets GMP standards, which provides superior production conditions for the production of glass containers and effectively avoids the impact of dust and bacteria on product cleanliness. The inspection system with 7 cameras and 360 continuously rotating and shooting, to detect the glass container body,mouth, shoulder, bottom,inner diameter,edge blind area and mouth bottom edge, as well the dimension, objects inside the container, cosmetic defects (bubbles, impurities and stones).The unqualified products such as crooked container are automatically eliminated, with a detection accuracy of 0.02mm, which ensures that the quality of the glass containers can reach the domestic YBB standard and the international ISO standard. "Excellence Centre"is a project established by Triumph Group and the world leading pharmaceutical packaging material manufacturer-Stevanato Group(Italy). The goal is to introduce world standard tubular glass containers production technology, technical process and quality standards to the Chinese market and make our company's products to keep up with international top standard.

Nationwide Seven Production Bases to Meet the Customers' needTriumph Junheng Co.,(hereinafter referred to as "Triumph Junheng") is a state-owned andprivate mixed ownership enterprise, Triumph Group of CNBM is the controlling shareholder.It ismainly engaged in the research and development, producing and sales of neutral Borosilicateglass tubing(hereinafter referred to as "Neutral Borosilicate glass tubing") and convertingtubular glass products. It has seven production bases located in Handan, Qingdao, Chongqing,Puyang, Anyang, Nanchang and Zhenjiang, which are expected to produce totally 170,000 tons of glass tubing and 136,000 tons of tubular glass container when they are reach full capacity. It is the first high-tech company which realizes stable mass production of high-quality Neutral borosilicate glass tubing with its "oxygen combustion + electric boosting"melting and Danner Tubing drawing technical process in China. It has leading R&D capabilities in the domestic pharmaceutical glass packaging industry, and can provide professional and personalized solutions of pharmaceutical glass packaging materials for materials for tubular glass converting and pharmaceutical companies.

Triumph Junheng builds a comprehensive research and development center for pharmaceutical neutral borosilicate glass in accordance with CNAS certification specifications. Continuously improve the level of laboratory quality management, minimize product quality risks, and meet the requirements of customers and regulations for the authenticity and reliability of testing/calibration to the greatest extent. The R&D center has a team with 30 people, covering various key technical fields such as raw materials analyzing, batching, melting, forming,and post-processing. The center has a building area of 800 square meters and consists of a precision instrument room, a physics laboratory, a chemical laboratory, a balance room, and a high temperature laboratory., Reagent room, equipped with 30 sets(sets)of R&D equipment such as atomic absorption spectrometer, expansion coefficient meter, and electron microscope. The R&D
center focuses on new technologies, intelligence, low energy consumption, localization and standardization, and actively undertakes national and local R&D projects to continuously achieve new technological breakthroughs.

Companys proucts have passed the Qulity inspection and Testing conducted by the national authoritative inspection agencies: Beijing Pharmaceutical Packaging Materials Inspection Institute and International Third Party Testing Agency-SGS. The product quality is fully in line with EP (European Pharmacopoeia), USP United States Pharmacopoeia),JP (Japanese Pharmacopoeia),According to the standard requirements of ISO12775 and the standard of "Neutral Borosilicate Glass Tubing" with Code YBB0012005-2-2015 approved by The National Food and Drug Administration. The expansion coefficient, boron content and hydrolitic resistance of products all have reached the international advanced level.

Quality Inspection
The international advanced automatic online inspection system has been adopted in the production line, which can automatically eliminate unqualified products with defects of dimension and cosmetic and unaligned ends treatment.
Automatic Packaging System
The automatic packaging system can complete the work of heat-shrinking into bundles and stacking into trays, effectively avoiding cosmetic defects such as scratches and impacting caused
by manual operation, ensuring the fastening of each bundle of products and the neatness of each tray.
- Q: How do you make the glass, please? What is the raw material?
- You can melt the sand and then try to remove the magazine and turn it into glass.
- Q: Is glass a recyclable resource?
- Building brick: using waste glass as a raw material, adding amount of the plasticity of clay material, pelletizing, forming, sintering, sintering at a temperature of 950 to 1050 DEG C for 180 210min can be prepared the production of building brick, the product with alkali resistance, high strength, not easy to fade and aging etc..
- Q: How can the invisible gauze of screen window increase its pressure resistance (glass fiber material)?
- To have good quality, if the quality of other yarn nets, there may be broken.
- Q: What's good with a cup?
- Is crisp or more toughness (generally good, there is a certain toughness), that is, to see whether it will be smashed at once, this kind of cup can not be.
- Q: What are the raw materials for low dielectric glass fiber?
- Glass fiber is divided into different grades according to its composition, properties and uses. According to the provisions of the standard level (see table), E glass fiber is most widely used, widely used in electrical insulating materials; special fiber grade S, while the output is small, but very important, because it has super strength, mainly used for military defense, such as bullet proof box; C level more than E with chemical resistance in the battery isolation plate, chemical filter; a basic glass fiber reinforced materials used in the production.The main component of glass fibre and its main components are silica, alumina, calcium oxide, boron oxide, sodium oxide, Magnesium Oxide, depending on the amount of alkali content in glass, can be divided into non alkali glass fiber (Na2O 0% ~ 2%, which belongs to the aluminum borosilicate glass), glass fiber (8% ~ sodium sodium calcium silicate glass 12%, a boron containing or not containing boron) and high alkali glass fiber (Na2O 13%, sodium calcium silicate glass).
- Q: What kind of mineral is glass made of?
- Photochromic glass (an advanced colored glass with an oxide of rare earths as a coloring agent)
- Q: Who can tell me the raw material and function of the flashy glass?
- Float and other molding methods, its advantages are: suitable for efficient manufacturing quality of flat glass, such as no wave, uniform thickness, smooth surface, upper and lower are parallel to each other; the scale of the production line is not affected by the forming method, energy consumption per unit of product is low; the finished high utilization rate; easy to scientific management and implementation across the board mechanization, automation, high labor productivity; continuous operation cycle of up to several years, is conducive to stable production; to provide suitable conditions for the on-line production of some new varieties, such as electric float reflective glass, annealing, cold end spraying film glass surface treatment etc..
- Q: Does the material of sunglasses choose good glass or plastic?
- Now, the most common use of sunglasses is resin lenses. The resin is a phenolic structure of chemicals, characterized by its light, high temperature resistance, impact resistance, and the ability to effectively block ultraviolet light. It should be said that this is a more excellent sunglasses, lens materials, polarized lenses made of this material will see more clearly.
- Q: What kind of film is toughened glass film?
- Tempered film is to prevent the phone dropped, smashed the screen. Not to prevent scratching. Ordinary film is the hardness of 3H, the use of a few months, there will not be too much scraping, the reason why the election of the steel screen is: high hardness, low toughness, mobile phones fell, can be very good anti broken screen. When the mobile phone drops on the ground, the impact is great, the tension is too large and the screen is broken. Toughened film toughness is low, when the handset sends out the tension, the membrane can bear the tension, thus greatly reduced the main screen to bear the tension.
- Q: Use of glass flake cement
- Coatings containing glass flakes are much harder than epoxy or polyester resin without glass flakes, and therefore have higher surface abrasion resistance.
Send your message to us
Ampoule ISO Standard YBB Standard High Quality
- Supplier:
 Triumph Junheng Co.,Ltd
Triumph Junheng Co.,Ltd
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- 100000 pc
- Supply Capability:
- 1000000000 pc/month
OKorder Service Pledge
OKorder Financial Service
Similar products
Hot products
Hot Searches






















