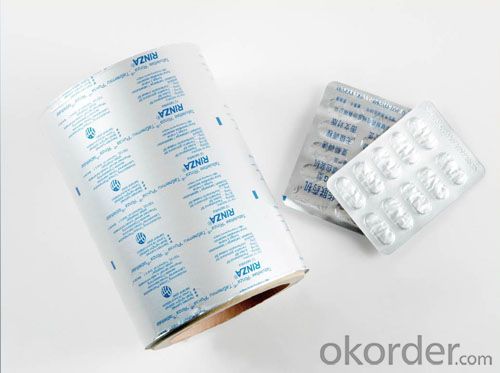
Aluminum Foil Tin Foil 8011 Alloy Foil for Pharmaceutical & Blister Packing
- Loading Port:
- China main port
- Payment Terms:
- TT OR LC
- Min Order Qty:
- 3 m.t.
- Supply Capability:
- 10000 m.t./month
OKorder Service Pledge
OKorder Financial Service
You Might Also Like
Specification
1.Description of Alloy 8011 Foil Printing Use For Pharmaceutical & Blister Packing Foil
1.structure:op/printing/AL/vc primer/AL/vc
2.thickness:0.012mm-0.036mm
3.width :60mm-780mm
4.Aolly:8011 H18
5.Usage: pharmaceutical blister packaging,heat seal against PVC ,PVDC
6.adopt ISO9001:2000 and SGS KOSHER MSDS
7:can be printed eight colors
2.Why you want to choose us?
We've been specialized in aluminium foil for more than ten years, we know this product very well, we know what is good, what is the market price.
3.Specification and Application of Alloy 8011 Foil Printing Use For Pharmaceutical & Blister Packing Foil
Product Name | Pharmaceutical Aluminium Blister Foil |
Color | silver, gold(as customer's requirement) |
Alloy | 8011 |
Temper | Half hard |
Thickness | 0.012mm-0.036mm |
Width | 60mm-780mm |
Core ID | 76mm |
quality tolerance | ±5% |
Structures | Primer/AL/VC OP/Printing/AL/VC |
Functions | High obstruction, Heat insulation,Printable,Breakable in slightly |
Certification | ISQ9000,SGS,MSDS,KOSHER |
MOQ | 500KG |
Payment | TT, L/C |
Delivery Time | 30-40 days after advanced payment |
Packing | wooden box/pallet as your request |
Applications | Blister packages for pharmaceutical products such as capsules and tablets,pill,pastille, Small-size food, candy, etc, heat seal against PVC ,PVDC |
4.Pictures of Alloy 8011 Foil Printing Use For Pharmaceutical & Blister Packing Foil

5.FAQ:
Q: Can you provide free samples?
A: Yes, free samples will be sent to you on freight at destination.
Q: Can I get your latest catalogue?
A: Yes, it will be sent to you in no time.
Q: What is the MOQ?
A: 5 tons
Q: What are your payment terms?
A: We accept L/C, T/T, etc.
If you are interested in our products, please do not hesitate to contact with us via following information
- Q: I'm bloody confusedCan someone help me with this problem?How much heat would be required to increase the temperature of 1.000 kg of aluminum by 4.000 degrees celsius?My chemistry teacher was gone all last week so this wasn't explained.
- This is an energy questionIt takes energy to raise the temperature of somethingFirst of all we need to know the heat capacity of aluminumYou should have some sort of table to find thisOne value I found was 0.897 J/(g-K) meaning it takes 0.897 Joules of energy to raise the temperature of 1 gram of aluminum by 1 degreeIn this case you have 1 Kg of aluminum, which is 1000 grams and you want to raise the temp by 4 degreesJ 0.897J/(g-K) x 1000g x 4K The g and K cancel out leaving J Do the mathGood luck!!
- Q: Is it possible to use aluminum foil for making a grilled cheese sandwich?
- <p>Yes, aluminum foil can be used to make a grilled cheese sandwich, although it's not the most common method. To do this, place the sandwich between two sheets of aluminum foil, ensuring the foil is large enough to completely wrap the sandwich. Seal the foil well to create a pouch. Then, place the foil-wrapped sandwich on a preheated grill or in an oven-safe pan on medium heat. Cook until the cheese is melted and the bread is toasted, checking frequently to prevent burning. Remember to be cautious when unwrapping the hot foil.</p>
- Q: I have a 2008 Dodge Sprinter and need to know where the Fluid goesOr if it even requires fluidIt makes a terrible noise when i turn the wheel.
- Usually there is no need to remove the old insulation; you can simply blow it right over the existingWe prefer cellulose insulation for applications such as theseCellulose has additives that make it perfectly safe and fire retardantCellulose creates a draft stopping effect, and has a slightly higher “R” value per inch than fiberglass insulation Fiberglass is perfectly acceptable but our preference is the cellulose because of the air blocking it achieves from densityThe biggest danger of blowing new insulation over the old is probably old style non-insulated rated can lightsIf these are present, you will need to construct a box to keep the insulation from making contact with the cansLook on the fixture for a sticker that says “IC RATED” and if that is not present then insulation cannot come into direct contact with the fixture Also keep in mind that you must be sure not to block off any attic ventilation points when blowing-in new insulation.
- Q: I'm going out of the country and I don't want my modelling clay to dry out.How can i store it so it doesn't dry out.
- Your kitty is probably going to end up sleeping with you! They like to stay warm and snuggle particularly during the cold monthsMine is constantly in my lap (like right now) or my shoulder, and sleeps between my legs at nightI would not spend money on a cat bed, I think they are unlikely to use it.
- Q: to come over and cook me some mac and cheese?
- You put the can of condensed milk (sealed) into a pot of water and boil it for 2 hoursI was skeptical about this myself but trust me, it worksAnd then when you open the can you have caramel
- Q: I'm looking to try and remake tandoori in my house, but I don't have a tandoori/clay ovenIs there any way to get the same effect while cooking at home, on perhaps the grill?
- A whole chicken will cook in 1 hour at 400 degreesGet yourself a thermometer, available at the groceryThe chicken is done when it registers 170 at the breast, close to the thighLet it sit, loosely covered with foil, for at least 10 minutes Chicken pieces cook in about half the time a whole chicken takesSo, 400 degrees for 30 minutesYou might try buying a pre-brined chicken or brining it yourselfJust soak the chicken or pieces in salt water overnight in your refrigerator before cookingIt helps keep it from drying outFor extra brown, crispy skin, pat it dry and rub it with olive oil before baking You really must have a thermometer, thoughIt also helps with cooking beef, pork and lamb.
- Q: So over the weekend I made roast beef and it came out ok but not perfect.it wasn't pink on the inside - which for any roast beef lover IS A BIG DEAL- so I guess it was overcookedBased on the weight (i don't remember now how much it weighed) the overseeing butcher/meat guy told me to cook it at 350 for about 1 hour, check on it 45 min into the cooking, and then take it out and cover w/aluminum foil so it continues to cookYou figure he'd know, right? Well I followed directions meticulously but when I went to check on it, after an hour it was still pretty much raw on the outsideSo I let it cook some more and wound up with an overcooked roast beefAny suggestions? Is there some infallible formula to go by? I'd like to make it for a dinner party but then it would probably HAVE TO BE PERFETTO - well it would be nice, at least, if it wereThanks!
- Well most people won't agree with ne but I know what I like I personally would not have opted for brisketTopside, sirloin or rib would be preferredBrisket needs longer cooking than other jointsin the ovenBut that is where i differAfter many years of roasting joints with varying results i bought a ceramic crock pot slow cookerI put the meat with an inch or so of water in the crock and crumble an oxo cube over itput on the lid and let it cook for 5 or 6 hours, very slowlyDo the veg as usual Again I prefer a steamer for the veg, except the potatoes I peel and cut potatoes into medium size pieces and part boil for 12 to 15 minutesA roast tin with hot oil and fat in the oven ,Drain the potatoes and fluff them with a forkPut in the roasting tin for about 25 minutes till browning(oven at a high temperature) At the same time you can be baking Yorkshire puddings., Remove beef from slow cooker and add stock or hot water and Bisto or whatever and make your gravy with the fluid from the slow cookerThe beef will be very tenderCarve and serve with the vegroast potatoes, Yorkshire and gravyAbsolutely deliciousWell that's the way I do it .
- Q: well, today i skipped lunch because i decided to work on some homework instead, so i had left my turkey and cheese sandwhich with mayonaise in my lockerI already left school and i just remeberedThe sandwhich has been there since 7:20 this morning, if i get to it by 7:20 tomorrow morning do you think it would have already started to smell bad enough for other people to smell through my locker? It's just that it's happened to me once before and it was really embarassing and i'm afraid it;; happen again(the sandwhich is rapped in aluminum foil and in a brown paper bag) thank you!
- There is a thin protective layer of insoluble aluminium oxide on all samples of the metalThis tends to slow down further oxidation.
Send your message to us
Aluminum Foil Tin Foil 8011 Alloy Foil for Pharmaceutical & Blister Packing
- Loading Port:
- China main port
- Payment Terms:
- TT OR LC
- Min Order Qty:
- 3 m.t.
- Supply Capability:
- 10000 m.t./month
OKorder Service Pledge
OKorder Financial Service
Similar products
Hot products
Hot Searches
Related keywords