Low Sulphur Calcined Petroleum Coke S 0.7
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- 11 m.t.
- Supply Capability:
- 10000000 m.t./month
OKorder Service Pledge
OKorder Financial Service
You Might Also Like
1.Structure of Calcined Petroleum Coke Description
Calcined Petroleum Coke is made from raw petroleum coke,which is calcined in furnace at a high temperature(1200-1300℃).CPC/Calcined Petroleum Coke is widely used in steelmaking,castings manufacture and other metallurgical industry as a kind of recarburizer because of its high fixed carbon content,low sulfur content and high absorb rate.Besides,it is also a best kind of raw materials for producing artifical graphite(GPC/Graphitized Petroleum Coke) under the graphitizing temperature(2800℃).
2.Main Features of the Calcined Petroleum Coke
High-purity graphitized petroleum coke is made from high quality petroleum coke under a temperature of 2,500-3,500°C. As a high-purity carbon material, it has characteristics of high fixed carbon content, low sulfur, low ash, low porosity etc.It can be used as carbon raiser (Recarburizer) to produce high quality steel,cast iron and alloy.It can also be used in plastic and rubber as an additive.
3. Calcined Petroleum Coke Images


4. Calcined Petroleum Coke Specification
PARAMETER UNIT GUARANTEE VALUE | ||
F.C.% | 98.5MIN | 98.5% MIN |
ASH % | 0.5 MAX | 0.8MAX |
V.M.% | 0.7 MAX | 0.7 MAX |
SULFUR % | 0.5 MAX | 0.7 MAX |
MOISTURE % | 0.5MAX | 0.5MAX |
SIZE | 0-5MM OR AS REQUEST | |
5.FAQ of Calcined Petroleum Coke
1). Q: Are you a factory or trading company?
A: We are a factory.
2). Q: Where is your factory located? How can I visit there?
A: Our factory is located in ShanXi, HeNan, China. You are warmly welcomed to visit us!
3). Q: How can I get some samples?
A: Please connect me for samples
4). Q: Can the price be cheaper?
A: Of course, you will be offered a good discount for big amount.
PARAMETER UNIT GUARANTEE VALUE | ||
F.C.% | 98.5MIN | 98.5% MIN |
ASH % | 0.5 MAX | 0.8MAX |
V.M.% | 0.7 MAX | 0.7 MAX |
SULFUR % | 0.5 MAX | 0.7 MAX |
MOISTURE % | 0.5MAX | 0.5MAX |
SIZE | 0-5MM OR AS REQUEST | |
- Q: How does carbon affect the formation of heatwaves?
- Carbon dioxide (CO2) and other greenhouse gases trap heat in the Earth's atmosphere, leading to a phenomenon known as the greenhouse effect. Increased carbon emission from human activities, such as burning fossil fuels, contributes to the rise in atmospheric CO2 levels. This, in turn, intensifies the greenhouse effect, causing global temperatures to rise. Heatwaves are extreme weather events characterized by prolonged periods of excessively hot weather. The increased concentration of carbon in the atmosphere contributes to the overall warming of the planet, making heatwaves more frequent, intense, and longer-lasting. Hence, carbon plays a significant role in the formation and exacerbation of heatwaves.
- Q: What are the health effects of carbon monoxide poisoning?
- Carbon monoxide (CO) poisoning can have severe health effects. When inhaled, carbon monoxide displaces oxygen in the bloodstream, leading to oxygen deprivation in vital organs. Symptoms range from mild, such as headaches and dizziness, to more severe, including confusion, loss of consciousness, and even death. Prolonged exposure to high levels of carbon monoxide can result in long-term neurological damage or cardiac complications. Therefore, it is crucial to have carbon monoxide detectors in homes and ensure proper ventilation to prevent poisoning.
- Q: What is carbon neutral shipping?
- The concept of carbon neutral shipping involves offsetting or balancing the carbon emissions produced during the transportation of goods by sea, air, or land. Its goal is to minimize the environmental and climate impact of shipping. Shipping contributes to greenhouse gas emissions by burning fossil fuels, primarily heavy fuel oil in ships' engines. This releases carbon dioxide (CO2), nitrogen oxides (NOx), sulfur oxides (SOx), and particulate matter into the atmosphere, contributing to global warming and air pollution. To achieve carbon neutrality in shipping, different strategies can be used. One common approach is carbon offsetting, which involves investing in projects that remove or reduce an equivalent amount of CO2 from the atmosphere. This can include reforestation, renewable energy projects, or methane capture initiatives. By supporting these projects, shipping emissions are balanced out, resulting in a net-zero carbon footprint. Another way to achieve carbon neutrality is by using alternative fuels and energy-efficient technologies. Biofuels, hydrogen, and electric propulsion systems can significantly reduce or eliminate carbon emissions from ships. Optimizing shipping routes and vessel design can also reduce fuel consumption and emissions. Collaboration between shipping companies, governments, and international organizations is crucial to promote carbon neutral shipping. This includes setting industry-wide emission reduction targets, implementing stricter regulations, and providing incentives for sustainable practices. While carbon neutral shipping is a positive step towards addressing climate change, it should be seen as a transitional measure towards a fully decarbonized shipping sector. Continued research and development in clean technologies, along with the adoption of sustainable practices, are essential for long-term environmental sustainability in the shipping industry.
- Q: How does carbon impact the availability of sustainable agriculture practices?
- The availability of sustainable agriculture practices is affected by carbon in several ways. Firstly, climate change is contributed to by carbon emissions from human activities, such as burning fossil fuels and deforestation. This change in climate patterns can result in extreme weather events like droughts, floods, and heatwaves, which can have a negative impact on agricultural productivity. Furthermore, the excessive presence of carbon in the atmosphere adds to the greenhouse effect, trapping heat and increasing global temperatures. This rise in temperature can disrupt natural ecosystems and decrease the amount of arable land available for agriculture. It can also change precipitation patterns, causing water scarcity or excessive rainfall, both of which can hinder sustainable agriculture practices. Carbon also plays a role in the health and fertility of soil. Soils can absorb excessive carbon dioxide from the atmosphere, which leads to increased soil acidity. This acidification can lower soil pH levels, making it difficult for crops to absorb necessary nutrients. Additionally, high carbon levels can affect soil microorganisms, which are essential for nutrient cycling and maintaining soil fertility. However, carbon can also have positive effects on sustainable agriculture practices. Carbon sequestration, the process of capturing and storing carbon dioxide from the atmosphere, can be utilized to improve soil health. Practices such as planting cover crops, adopting agroforestry systems, and implementing no-till farming techniques can help sequester carbon in the soil, enhancing its fertility and ability to withstand challenges. This, in turn, promotes sustainable agriculture by increasing crop yields, reducing the need for synthetic fertilizers, and improving the soil's ability to retain water. In conclusion, carbon emissions and their impact on climate change and soil health have a significant influence on the availability of sustainable agriculture practices. It is crucial to mitigate carbon emissions and adopt practices that sequester carbon in order to ensure a sustainable and resilient agricultural system in the face of climate change.
- Q: How are carbon compounds classified?
- Carbon compounds are classified based on their structural arrangement, functional groups, and the type of bonds they form with other elements.
- Q: Recently bought an alarm clock, it is recommended to use carbon batteries. Nanfu battery is not good for the movement.
- Yes, a lot of people do not pay attention to, the Nanfu battery, strange carbon batteries say it is really rare, generally small supermarkets are not only basic, and is strongly alkaline. Therefore, it is best to go to a large supermarket or electrical store try, there are generally sold there, and many types, the choice will be more.
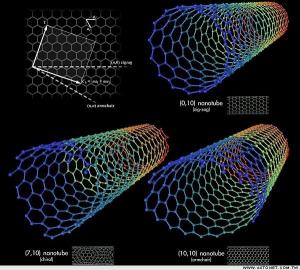
- Q: What are the properties of carbon nanotubes?
- Cylindrical structures made entirely of carbon atoms are known as carbon nanotubes. They possess a distinct set of properties that make them highly sought after in various fields of science and technology. Some of the notable properties of carbon nanotubes are as follows: 1. Remarkable strength and stiffness: Carbon nanotubes have an exceptional strength-to-weight ratio, making them one of the strongest materials discovered so far. They are approximately 100 times stronger than steel, yet significantly lighter. This characteristic renders them suitable for applications requiring lightweight materials with high strength. 2. Excellent electrical conductivity: Carbon nanotubes exhibit excellent electrical conductivity, enabling efficient flow of electrical current. They can be utilized as conductive components in diverse electronic devices, including transistors, sensors, and energy storage systems. 3. Efficient thermal conductivity: Carbon nanotubes possess high thermal conductivity, allowing efficient heat transfer. This property makes them ideal for applications requiring effective dissipation of heat, such as thermal management in electronic devices. 4. Flexibility and resilience: Carbon nanotubes are highly flexible and can endure substantial deformation without fracturing. They can be bent and twisted without compromising their structural integrity, making them suitable for applications demanding flexibility, such as flexible electronics. 5. Unique optical and mechanical properties: Carbon nanotubes possess distinctive optical properties that vary depending on their structure and arrangement. They can absorb and emit light across a wide range of wavelengths, making them valuable in applications like photodetectors and solar cells. Additionally, their mechanical properties, including elastic deformation, contribute to their usefulness in applications requiring shock absorption and impact resistance. 6. Chemical stability: Carbon nanotubes exhibit high chemical stability, enabling them to resist degradation or corrosion when exposed to different chemical environments. This characteristic makes them suitable for applications in harsh conditions or as protective coatings. 7. Large aspect ratio: Carbon nanotubes possess a high aspect ratio, with lengths often exceeding thousands of times their diameter. This characteristic allows them to form robust and lightweight composite materials when integrated into a matrix, enhancing the overall strength and stiffness of the composite. In conclusion, the combination of properties displayed by carbon nanotubes makes them an intriguing and versatile material with enormous potential in various applications, including electronics, aerospace, medicine, and energy storage.
- Q: What are the effects of carbon emissions on freshwater systems?
- Carbon emissions have significant effects on freshwater systems. Increased carbon dioxide levels in the atmosphere lead to a rise in global temperatures, which in turn affects freshwater ecosystems. Warmer water temperatures can disrupt the delicate balance of aquatic life, leading to the decline of certain species, including fish and other organisms that depend on specific temperature ranges. Additionally, carbon emissions contribute to ocean acidification, which ultimately affects freshwater systems through interconnected hydrological cycles. Acidic waters can harm freshwater organisms, deplete their food sources, and disrupt the overall health of these ecosystems. Overall, carbon emissions have a detrimental impact on freshwater systems, threatening their biodiversity and ecological stability.
- Q: How does carbon dioxide affect the Earth's atmosphere?
- Carbon dioxide affects the Earth's atmosphere by trapping heat from the sun, leading to the greenhouse effect and causing global warming and climate change.
- Q: What are the economic impacts of carbon emissions?
- Carbon emissions have wide-ranging and significant economic effects. These emissions, primarily from burning fossil fuels, contribute to climate change and global warming. The resulting climate changes directly impact various economic sectors and can have both short and long-term economic consequences. Dealing with the effects of climate change is one of the most noticeable economic impacts of carbon emissions. As a result of these emissions, extreme weather events like hurricanes, floods, and droughts become more frequent and intense. These events cause extensive damage to infrastructure, homes, and businesses, resulting in significant economic losses. For instance, in 2017, the United States experienced a record-breaking hurricane season, with hurricanes Harvey, Irma, and Maria causing approximately $265 billion in damages. Furthermore, carbon emissions also affect agricultural productivity. Climate change alters temperature and precipitation patterns, disrupting crop production and reducing yields. This, in turn, impacts food prices and availability, affecting both consumers and farmers. Additionally, carbon emissions contribute to ocean acidification, which harms marine ecosystems and disrupts fisheries, leading to economic losses for fishing communities. Moreover, carbon emissions have implications for public health, creating economic burdens. Air pollution caused by carbon emissions can lead to respiratory and cardiovascular illnesses, increasing healthcare costs and reducing workforce productivity. Additionally, extreme heatwaves, exacerbated by carbon emissions, negatively impact worker productivity and labor capacity, affecting economic output. To mitigate the economic impacts of carbon emissions, many countries have implemented policies and regulations to reduce greenhouse gas emissions. These policies often involve carbon pricing mechanisms, such as carbon taxes or cap-and-trade systems, that aim to incentivize the transition to cleaner energy sources and decrease carbon emissions. While these policies may have short-term economic costs, they also create opportunities for innovation and the development of green technologies, leading to long-term economic benefits. In conclusion, the economic impacts of carbon emissions are significant and diverse. They range from the costs associated with climate-related disasters to the effects on agriculture, public health, and productivity. Addressing these impacts through effective climate policies is vital to mitigate economic risks and promote a sustainable and resilient economy.
Send your message to us
Low Sulphur Calcined Petroleum Coke S 0.7
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- 11 m.t.
- Supply Capability:
- 10000000 m.t./month
OKorder Service Pledge
OKorder Financial Service
Similar products
Hot products
Hot Searches
Related keywords