Amino Trimethylene Phosphonic Acid Water Treatment Chemical
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- 6000 m.t./month
OKorder Service Pledge
OKorder Financial Service
You Might Also Like
Product Description:
Amino tris(methylene phosphonic acid) / Amino Trimethylene Phosphonic Acid/ ATMP / 6419-19-8 / C3H12NO9P3
CAS No. 6419-19-8
Molecular Formula: N(CH2PO3H2)3
Molecular weight: 299.05
Structural Formula: 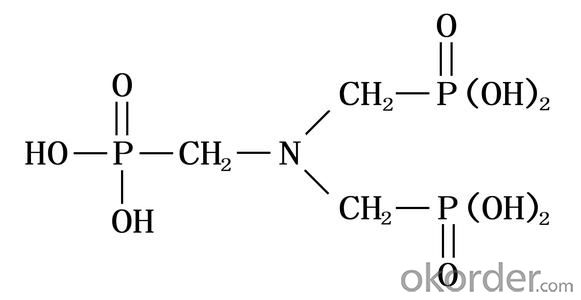
Properties:
ATMP has excellent chelation, low threshold inhibition and lattice distortion ability. It can prevent scale formation, calcium carbonate in particular, in water system. ATMP has good chemical stability and is hard to be hydrolyzed in water system. At high concentration, it has good corrosion inhibition.
ATMP is used in industrial circulating cool water system and oilfield water pipeline in fields of thermal power plant and oil refinery plant. ATMP can decrease scale formation and inhibit corrosion of metal equipment and pipeline. ATMP can be used as chelating agent in woven and dyeing industries and as metal surface treatment agent.
The solid state of ATMP is crystal powder, soluble in water, easily deliquescence, suitable for usage in winter and freezing districts. Because of its high purity, it can be used in woven & dyeing industries and as metal surface treatment agent.
Specification:
| Items | Index | |
|---|---|---|
| Standard | Solid | |
| Appearance | Clear, Colorless to pale yellow aqueous solution | White crystal powder |
| Active acid % | 50.0-51.0 | 95.0min |
| Chloride (as Cl-)% | 1.0 max | 1.0 max |
| pH value (1% solution) | 2.0 max | 2.0 max |
| Fe,mg/L | 10.0max | 20.0max |
| Density (20°C)g/cm3 | 1.31-1.35 | - |
| Colour APHA (Hazen) | 30.0max | - |
Application range&using method:
ATMP is usually used together with other organophosphoric acid, polycarboxylic acid and salt to built all organic alkaline water treatment agent. ATMP can be used in many different circulating cool water system. The recommended dosage is 5-20mg/L. As corrosion inhibitor, The recommended dosage is 20-80mg/L.
Package and Storage:
ATMP liquid: Normally In 30kg or 250kg net Plastic Drum;ATMP solid: 25kg inner liner polyethylene (PE) bag, outer plastic woven bag, or confirmed by clients request.Storage for ten months in room shady and dry place.
Safety Protection:
ATMP is Acidity, Avoid contact with eye and skin, once contacted, flush with water.
Shipping Date: Within 7-10 workdays after receiving your deposit.
Our Service:
Own Lab and joint venture factory.
Superb r&d team;Safety standardization production.
Rich experience in export and strong logistical support.
Good relationship with many large domestic pharmaceutical factory.
Perfect service, perfect supply chain.
- Q:What is the PTC catalyst in chemistry?
- PTC, Phase Transfer Catalyst, phase transfer catalyst PTC is the role of a reaction from one phase to another phase to participate in the reaction to promote a solvent can be dissolved in organic solvents and a solvent insoluble in the ion Reagents react between the two reagents. And this type of reaction is collectively referred to as phase transfer catalytic reaction.
- Q:Junior high school chemistry - chemical reaction before and after the quality and chemical properties of the material must be the catalyst?
- It is not always possible that the equivalent reaction, i.e. one or more of the reactants, is the same as the relative atomic mass of one or more of the products and the coefficients in the chemical equation are the same
- Q:What is the definition and function of the catalyst in chemistry?
- Definition: in the chemical reaction can change the chemical reaction rate of other substances, and its own quality and chemical properties before and after the reaction did not change the material called catalyst, also known as catalyst. The role of the catalyst in the chemical reaction is called catalysis. The use of a catalyst only changes the rate of chemical reaction and does not alter the quality of the product.
- Q:What is the meaning of catalyst in chemistry?
- In the chemical reaction can change the reaction rate of chemical reaction (increase or decrease) without changing the chemical balance, and its own quality and chemical properties in the chemical reaction before and after the material did not change the catalyst.
- Q:What kind of compounds or elements can be used as catalysts in high school chemistry? What is the catalyst for what?
- Iron, manganese dioxide, mainly metal and its oxides.
- Q:No one knows the expression of the catalyst and the chemical expression of the acridine
- If it is potassium permanganate oxygen, the catalyst is only manganese dioxide, so write on the horizontal line MnO2
- Q:Exemplify the use of green catalysts in green chemistry
- Catalyst does not participate in the reaction If you ask the green oxidizer, then there is hydrogen peroxide catalyst is the quality of reactants before and after the change
- Q:The catalyst before and after the reaction of the quality and nature of the same, does it mean that a little catalyst can be all the substrate reaction? Such as: one gram of MnO2 can reflect the infinite H2O2?
- Catalyst reaction is essentially involved in the reaction, we learn the chemical reaction is simplified, in fact, most of the reactions are to go through several processes to form, because it is more complex, we learn to write directly reactants and products , The reaction is saved in the middle of the catalyst is involved in the middle of the reaction process, in the final process and generated, to achieve the effect of catalytic reaction, its own quality and nature has not changed.
- Q:Now, i am studying for my biology exam in 3 weeks time...i stumbled upon catalase, and then checked my book its catalyst...now im confused...is there a different among these 2 terms? i think..catalyst is the when a substance brings up or about a chemical reaction without using itself up and then catalase breaks down the toxic by-product of metabolism, hydrogen peroxide, into water and oxygen.Or am i wrong?please explain what is catalyst and catalase in biology or are they the same, just differently?
- an enzyme that decomposes hydrogen peroxide into oxygen and water.a substance that causes or accelerates a chemical reaction without itself being affected. To put thing simply, a catalyst is any substance that speeds up a chemical reaction. These can be natural or manmade. Catalase is actually a specific type of naturally-occuring catalyst, an enzyme in cells that decomposes hydrogen peroxide (Which is extremely toxic to life!) into harmless components. Catalase enzymes are highly concentrated in the aptly named cell organelles known as peroxisomes. Just remember- if the word ends in -ase, it's a type of enzyme! :) Hope this information helps!
- Q:in my 99 ram 2500 v8 5.9 that code came up, not sure what to do about it or what it could be any ideas or help with be great thanks
- This Site Might Help You. RE: catalyst efficiency low bank 1? in my 99 ram 2500 v8 5.9 that code came up, not sure what to do about it or what it could be any ideas or help with be great thanks
1. Manufacturer Overview |
|
|---|---|
| Location | |
| Year Established | |
| Annual Output Value | |
| Main Markets | |
| Company Certifications | |
2. Manufacturer Certificates |
|
|---|---|
| a) Certification Name | |
| Range | |
| Reference | |
| Validity Period | |
3. Manufacturer Capability |
|
|---|---|
| a)Trade Capacity | |
| Nearest Port | |
| Export Percentage | |
| No.of Employees in Trade Department | |
| Language Spoken: | |
| b)Factory Information | |
| Factory Size: | |
| No. of Production Lines | |
| Contract Manufacturing | |
| Product Price Range | |
Send your message to us
Amino Trimethylene Phosphonic Acid Water Treatment Chemical
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- 6000 m.t./month
OKorder Service Pledge
OKorder Financial Service
Similar products
New products
Hot products
Related keywords


























