market price for carbon black /Rubber Antioxidant
- Loading Port:
- Qingdao
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- 20000 m.t./month
OKorder Service Pledge
OKorder Financial Service
You Might Also Like
Specifications
Rubber Antioxidant IPPD(4010NA)
1.101-72-4
2.Professional production factory
3.High quality
4.Used in the manufacture of tire
market price for carbon black /Rubber Antioxidant IPPD(4010NA)/ CAS No:101-72-4
(1)Product Name: Rubber Antioxidant IPPD (4010NA)
(2)Chemical Name: N-isopropyl-N'-phenyl-p-phenylene diamine
(3)Structure:
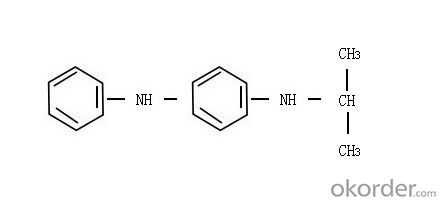
(4)Molecular Formula: C15H18N2
(5)Molecular Weight : 226.4
(6)CAS: 101-72-4
(7)Specification:
Index Name | Qualified Grade |
Appearance(eye measurement) | dark brown to dark violet pastilles |
Initial M.P.,°C ≥ | 70.0 |
Heating loss % ≤ | 0.30 |
Ash content % ≤ | 0.20 |
(GC)/Assay % ≥ | 95.0 |
Properties
Grayish purple to purple brown pastilles. Relative density is 1.14g/cm3. Soluble in benzene, acetone, ethyl acetate,
Carbon tetrachloride, chloroform, etc. Difficultly soluble in gasoline. Insoluble in water.
Application
It is an excellent and universal antioxidant used for natural rubber, synthetic rubber and latex. Better protective
performance of ozone, flex cracking, also the excellent protective agent for the heat, oxygen, light and the general
aging. It is able to inhibit harmful metals. It is mainly used in tires, rubber hose, belt and industrial rubber products.
Storage
The product should be sealed and stored in the dry, cool, well-ventilated area. Avoid exposure to direct
sunlight.
Packaging
25kg plastic woven bag lined with plastic film bag,kraft paper bag.
Following is our main products
Intermediate | Benzothiazole; MBT-NA | |
Rubber Accelerator | Thiazoles | MBT(M); MBTS(DM); ZMBT(MZ) |
Sulfonamides | CBS(CZ); MBS(NOBS); NS(TBBS); DCBS(DZ) | |
Dithiocarbamates | ZDEC(EZ); ZDMC(PZ); ZDBC(BZ) | |
Thiurams | TMTD(TT); TMTM(TS) | |
Guanidines | DPG(D) | |
Rubber Antioxidant | A(PAN); D(PBN); RD(TMQ); 4010(IPPD); 4020(6PPD); BLE | |
Antisorching Agent | PVI(CTP) | |
Rubber Vulcanizing Agent | DTDM; Insoluble Sulfur | |
Our Rubber Accelerator IPPD (4010NA) is produced by new technology---"enclosed clean
production techniques", located the forefront of industry in skill and output. Welcome to inquiry!
- Q:Palladium is the main catalyst in chemistry?
- Palladium in the chemical mainly to do the catalyst; palladium and ruthenium, iridium, silver, gold, copper and other alloy, can improve the palladium resistivity, hardness and strength, used in the manufacture of precision resistors, jewelry and so on. While the most common and most commercially available palladium jewelery is palladium.
- Q:Is the catalyst considered a chemical reaction?
- The middle school textbook defines that the catalyst itself does not participate in chemical reactions
- Q:Is palladium predominantly a catalyst in chemistry?
- Palladium in the chemical mainly to do the catalyst; palladium and ruthenium, iridium, silver, gold, copper and other alloy, can improve the palladium resistivity, hardness and strength, used in the manufacture of precision resistors, jewelry and so on.
- Q:Chemical equation if there is a catalyst and heating, which write in the equal sign above, which written in the following? Tomorrow academic level test, solution
- At the same time, the catalyst is heated
- Q:What is the definition and function of the catalyst in chemistry?
- Definition: in the chemical reaction can change the chemical reaction rate of other substances, and its own quality and chemical properties before and after the reaction did not change the material called catalyst, also known as catalyst. The role of the catalyst in the chemical reaction is called catalysis. The use of a catalyst only changes the rate of chemical reaction and does not alter the quality of the product.
- Q:How are the 4 characteristics of a catalyst (1. organic or inorganic 2. reusable 3. Highly specific, and 4. lowers activation energy) important in preforming life functions? please be as specific as possible, i understand that these are characteristic, i just don't understand why they're beneficial, other than the reusable and lowers activation energy one.
- Organic or Inorganic - the catalyst (enzyme) must be organic to be found in the cell. Catalysts speed up chemical reactions inside a cell and must therefore be organic to be a functioning part of the cell. Reusable - There are so many reactions that catalysts are involved in that it would be a waste for the cell if a catalyst could only last one reaction, especially if there are inhibitors and competition for the active site. Catalysts must be reusable in order to keep the cell functioning. Catalysts always remain unchanged after a reaction. HIihly Specific - Catalysts are only made to catalyze one specific chemical reaction. Their active site has proteins bonded in such a way that only certain elements can enter the active site and H bond with those proteins. The fact that they are highly specific maximizes the productiveness of the cell. And it ensures that the cell only has catalysts to reactions that it needs to be completed. It also ensures that the elements are correctly bonded with eachother. If any two elements could enter the active site, there is no guarantee that the correct product will be produced. Catalysts and Enzymes must be super highly specific in order to properly function. Lowers Activation Energy - The more energy a cell has to spend to catalye a reaction, the worse it is for the cell and the less ATP is has for other reactions. Catalyts hold the substrates together so there is less energy that is needed to have the two substrates react with eachother. Activation Energy is the energy that is needed to start a reaction. So the less energy used by the cell for reactions, the better for the cell. Hope this helps
- Q:What is the catalyst in the end?
- You said the chemical catalyst or Ati graphics card catalyst? If the above is enough to explain the above, if it is the latter, that is, the meaning of the graphics card, Ati's graphics drive like a catalyst
- Q:What is the difference between an enzyme catalyst in a living body and a catalyst in chemistry?
- enzyme susceptibility inactivation Chemical catalyst under certain conditions, due to poisoning and loss of catalytic capacity; and enzyme than the chemical catalyst is more fragile, more volatile activity. Any factor that makes the protein degenerate (to strengthen the acid, alkali, high temperature, etc.), can make the enzyme completely lost activity.
- Q:Why extract the genome, the digestion is always not cut
- The enzyme, like the general catalyst, only catalyzes the thermodynamics of the permissible chemical reaction, shortening the time to reach the chemical equilibrium without changing the equilibrium point. The enzyme as a catalyst has no qualitative and quantitative changes before and after the chemical reaction. The mechanism of action of enzymes and general catalysts is to reduce the activation energy of the reaction.
1. Manufacturer Overview |
|
|---|---|
| Location | |
| Year Established | |
| Annual Output Value | |
| Main Markets | |
| Company Certifications | |
2. Manufacturer Certificates |
|
|---|---|
| a) Certification Name | |
| Range | |
| Reference | |
| Validity Period | |
3. Manufacturer Capability |
|
|---|---|
| a)Trade Capacity | |
| Nearest Port | |
| Export Percentage | |
| No.of Employees in Trade Department | |
| Language Spoken: | |
| b)Factory Information | |
| Factory Size: | |
| No. of Production Lines | |
| Contract Manufacturing | |
| Product Price Range | |
Send your message to us
market price for carbon black /Rubber Antioxidant
- Loading Port:
- Qingdao
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- 20000 m.t./month
OKorder Service Pledge
OKorder Financial Service
Similar products
New products
Hot products
Hot Searches
Related keywords




























