Amino Tri Methylene Phosphonic acid ATMP
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- 6000 m.t./month
OKorder Service Pledge
OKorder Financial Service
You Might Also Like
Product Description:
Amino tris(methylene phosphonic acid) / Amino Trimethylene Phosphonic Acid/ ATMP / 6419-19-8 / C3H12NO9P3
CAS No. 6419-19-8
Molecular Formula: N(CH2PO3H2)3
Molecular weight: 299.05
Structural Formula: 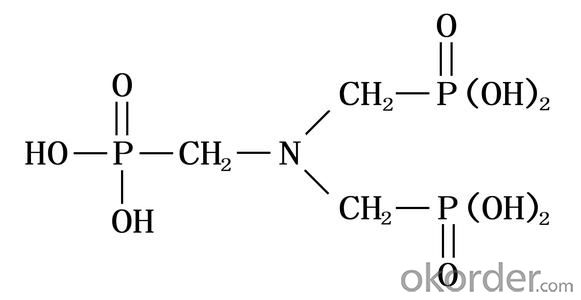
Properties:
ATMP has excellent chelation, low threshold inhibition and lattice distortion ability. It can prevent scale formation, calcium carbonate in particular, in water system. ATMP has good chemical stability and is hard to be hydrolyzed in water system. At high concentration, it has good corrosion inhibition.
ATMP is used in industrial circulating cool water system and oilfield water pipeline in fields of thermal power plant and oil refinery plant. ATMP can decrease scale formation and inhibit corrosion of metal equipment and pipeline. ATMP can be used as chelating agent in woven and dyeing industries and as metal surface treatment agent.
The solid state of ATMP is crystal powder, soluble in water, easily deliquescence, suitable for usage in winter and freezing districts. Because of its high purity, it can be used in woven & dyeing industries and as metal surface treatment agent.
Specification:
| Items | Index | |
|---|---|---|
| Standard | Solid | |
| Appearance | Clear, Colorless to pale yellow aqueous solution | White crystal powder |
| Active acid % | 50.0-51.0 | 95.0min |
| Chloride (as Cl-)% | 1.0 max | 1.0 max |
| pH value (1% solution) | 2.0 max | 2.0 max |
| Fe,mg/L | 10.0max | 20.0max |
| Density (20°C)g/cm3 | 1.31-1.35 | - |
| Colour APHA (Hazen) | 30.0max | - |
Application range&using method:
ATMP is usually used together with other organophosphoric acid, polycarboxylic acid and salt to built all organic alkaline water treatment agent. ATMP can be used in many different circulating cool water system. The recommended dosage is 5-20mg/L. As corrosion inhibitor, The recommended dosage is 20-80mg/L.
Package and Storage:
ATMP liquid: Normally In 30kg or 250kg net Plastic Drum;ATMP solid: 25kg inner liner polyethylene (PE) bag, outer plastic woven bag, or confirmed by clients request.Storage for ten months in room shady and dry place.
Safety Protection:
ATMP is Acidity, Avoid contact with eye and skin, once contacted, flush with water.
Shipping Date: Within 7-10 workdays after receiving your deposit.
Our Service:
Own Lab and joint venture factory.
Superb r&d team;Safety standardization production.
Rich experience in export and strong logistical support.
Good relationship with many large domestic pharmaceutical factory.
Perfect service, perfect supply chain.
- Q:Chemical "catalyst can speed up the chemical reaction rate of other substances," this sentence right?
- Wrong, the catalyst is divided into two kinds, one is to speed up the chemical reaction speed, and the other is the opposite
- Q:what is metallocene catalyst technology?
- Metallocene catalyst A transition-metal atom sandwiched between ring structures having a well-defined single catalytic site and well-understood molecular structure used to produce uniform polyolefins with unique structures and physical properties. See also Catalysis; Coordination chemistry; Coordination complexes; Metallocenes; Organometallic compound. In the early 1980s, W. Kaminsky discovered that an appropriate co-catalyst activated metallocene compounds of group 4 metals, that is, titanium, zirconium, and hafnium, for alpha-olefin polymerization, attracting industrial interest. This observation led to the synthesis of a great number of metallocene compounds for the production of polymers already made industrially, such as polyethylene and polypropylene, and new materials. Polymers produced with metallocene catalysts represent a small fraction of the entire polyolefin market, but experts agree that such a fraction will increase rapidly in the future. See also Polymer; Polymerization; Polyolefin resins.
- Q:I think doubling the concentration of catalyst will double the rate of a reaction (K2/K1 =2). I want to confirm. This is for my project work. Please reply as soon as possible.
- A catalyst speeds up a chemical reaction by providing an alternate reaction pathway with a lower activation energy, thus increasing the number of collisions that can result in the formation of product. When the catalyst is a reactant in the rate determining step, and the reaction is first order in the catalyst, then a doubling of the concentration will double the rate. But if the rate determining step which includes the catalyst is not first order, then doubling the concentration won't double the rate. Then there is the case of a heterogeneous catalyst in which the reaction is essentially zero order in the catalyst. The amount of catalyst won't affect the speed of the reaction beyond the initial increase. The mere fact that the catalyst is present speeds up the reaction.
- Q:What is the difference between a catalyst and an inducer in a chemical reaction?
- The catalyst does not participate in the reaction, but only the carrier of the reaction; the inducer will participate in the reaction
- Q:Why the catalyst is required to have a large surface area and a rich pore structure
- Has a large surface area, is the reactants and catalyst contact more fully, improve the catalytic effect
- Q:Does the nature and quality of the catalyst itself change before and after the chemical reaction?
- The catalyst is not consumed in the chemical reaction. They are able to be separated from the reactants, either before or after the reaction. However, they may be consumed at a certain stage of the reaction and then regenerated before the end of the reaction.
- Q:Also, how is the catalyst affected by heat? Please answer all of the questions not just one of the three. THANK YOU!
- It denatures the catalyst because the rise in pH or amount of H3O+ ions. Temperature will also denature the catalyst if it's out of its optimum range.
- Q:how a catalyst can provide a new route in forming the product?
- a catalyst does not provide a new route. it just lowers something called the energy of activation of the reaction. this makes some changes in the thermodynamics and kinetics of the reaction
- Q:Does increasing the amount of catalyst added to, say, a solution of Hydrogen Peroxide, make the rate of reaction go faster. Is the rate of reaction directly proportional to the amount of catalyst added to the solution? Or does the experiment go at the same rate regardless of how much catalyst there is? Thanks would really appreciate some answers. - Sarah
- Adding a catalyst would increase the rate of reaction. This could decrease the activation energy, the amount of kinetic energy needed for the reaction to occur. Hope that helps
- Q:What is the effect of the catalyst in chemistry?
- Changing the rate of reaction can speed up the reaction and slow down the rate of reaction
1. Manufacturer Overview |
|
|---|---|
| Location | |
| Year Established | |
| Annual Output Value | |
| Main Markets | |
| Company Certifications | |
2. Manufacturer Certificates |
|
|---|---|
| a) Certification Name | |
| Range | |
| Reference | |
| Validity Period | |
3. Manufacturer Capability |
|
|---|---|
| a)Trade Capacity | |
| Nearest Port | |
| Export Percentage | |
| No.of Employees in Trade Department | |
| Language Spoken: | |
| b)Factory Information | |
| Factory Size: | |
| No. of Production Lines | |
| Contract Manufacturing | |
| Product Price Range | |
Send your message to us
Amino Tri Methylene Phosphonic acid ATMP
- Loading Port:
- Tianjin
- Payment Terms:
- TT OR LC
- Min Order Qty:
- -
- Supply Capability:
- 6000 m.t./month
OKorder Service Pledge
OKorder Financial Service
Similar products
New products
Hot products

























